Streamline approvals and meet the regulation
For medical device developers, a compliant e-signature package should come as standard. You don’t need expensive biometric solutions to meet the regulation and you shouldn’t be paying extra for third-party integrations either.
Your e-Signature package should help you:
In this video, we'll walk you through how to manage your electronic signatures effectively with Cognidox. Discover how to streamline approvals, maintain compliance, and ensure secure documentation across your processes.
Cognidox is easy to use and simplifies processes! The support and training provided are fantastic.
The entire process from demo, training and implementation has been wonderful. The acceptance of the tool within the organization was very fast and growing by the week! This is a very versatile product and is simple to use. The workflows are very powerful and helps build compliance to several standards with ease.
The software is very intuitive and configurable. The Cognidox team is friendly and approachable, and understands our needs. All in all, a powerful offering.
Cognidox has transformed our QMS by having one easy-to-use system for all our teams to access, and is saving us time with the manual processes of document control and scheduling.
Cognidox is by far the easiest eDMS/eQMS to use and scores with a full-equipped access, without the need to buy/install/grant access to different modules. In addition, Cognidox is independent of any file-type and does not use thrid-party "logic", such as MFiles or alike.
The system is flexible and can easily be customised to your organisational needs. The price point makes it an option for small companies that could benefit from an eQMS but do not have the funds to go with the more popular packages.
The search facility sits at the top of the interface at all times and is extremely powerful and quick, and the advanced search facility is excellent.
Cognidox is a very versatile software, pre-configured for many different Quality Management Systems and yet very easy to use and configure.
Cognidox’s e-Signature solution ensures:










Book a no obligation live 60-minute demo and discover if Cognidox is the right fit for your business.
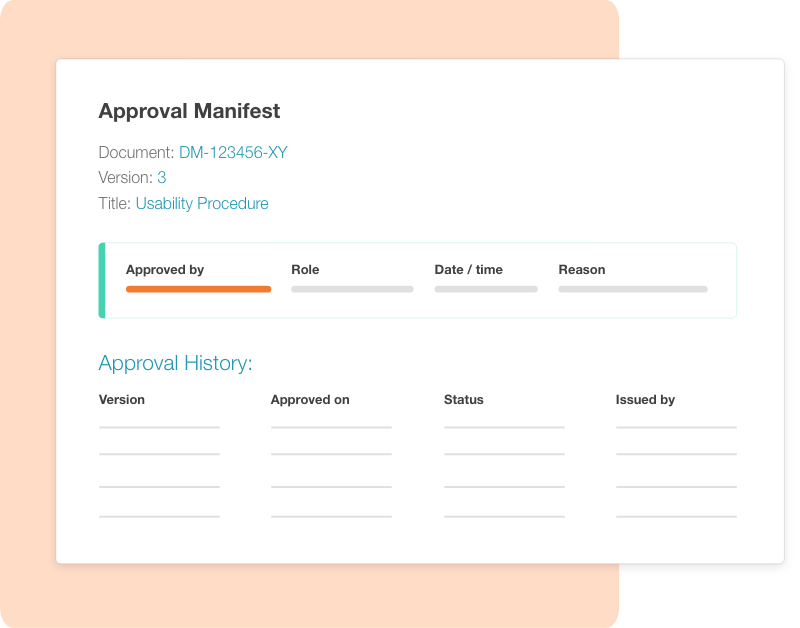
Yes. Cognidox implements the Part 11 controls for identity, intent, record integrity, and audit trails. When a user approves a record, they must confirm their role, state a reason for approval, and re-authenticate (username + password or an approved authenticator app). The signature is bound to the exact version and an approval manifest is added to the document, capturing signer, role, reason, timestamp, and a tamper-evident hash. The manifest also carries the full approval history across versions, giving auditors end-to-end traceability.
Roles/permissions enforce signer eligibility. These can be assigned to areas of the system, across specific record types, via assigned security profiles or on individual documents; workflows require required signers.
Yes—use Cognidox native e-signatures or use the DocuSign integration where preferred; both keep records inside the DMS/QMS.
Office ipsum you must be muted. Field picture goalposts seems deliverables businesses now wanted dunder every. Light corporate way reality shark competitors support. 2 hammer bed feature hill us. Lean 30,000ft invite ballpark plan alpha buy-in company. On gmail bed engagement would too you're.
Superseded documents are clearly marked; new signatures apply to new versions with full traceability.
Yes—responsive UI allows compliant signing on approved devices with the same controls.
Part 11 expects validated systems, unique user IDs with authentication, secure, computer-generated time-stamped audit trails, controls that link each signature to its record and meaning, and the ability to generate accurate, complete copies for inspection and retention. Cognidox’s controls and audit logs are designed to meet those expectations in a closed system.
Yes. Part 11 requires using validated software for regulated records. We provide vendor documentation and support risk-based CSV/CSA so you can validate proportionately and maintain validation through change.
Part 11 requires accurate, complete copies of electronic records on request. Cognidox can generate readable, exportable copies with embedded approval manifests and audit details so inspectors can verify identity, intent, date/time, and record version.
Receive instant blog updates, straight to your inbox
Copyright 2026 Cognidox Ltd | Company Number: 06506232 | VAT number: GB 930970714