- What is a medical device
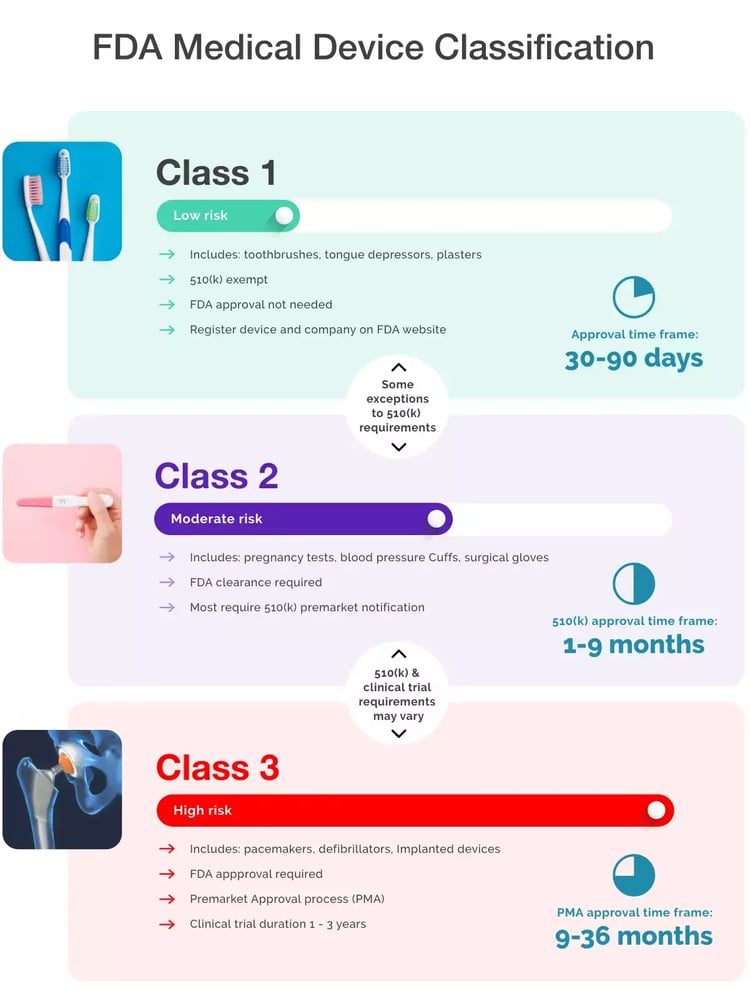
- Regulations in the EU, the UK and the US
- The QMS: controlling the development process
- The importance of training in medical device development
- Risk management
- Design, verification and validation
- Managing quality events
- Prepare your technical documentation for audit
- Medical device development - what do you really need?
The complete guide to medical device development
If you want to be a successful medical device developer, you need more than just a great idea for a product and skills to build it. You also need rigour and control.
This page is a guide to designing and developing a medical device in a fast-moving and disrupted marketplace.
The scale of the medical device development challenge
Medical device development is one of the most heavily regulated sectors in the world. The consequences and penalties for making mistakes and releasing unsafe or poorly designed devices can be serious. Meanwhile, the distinction and boundaries between software, apps and medical devices are blurring. They’re presenting new opportunities and complexities for developers.
With more cutting-edge tech on the horizon, the rewards for success are considerable:
.webp?width=750&name=med-dev-intro-info-cropped-2%20(1).webp)
As a developer, therefore, you need to make sure you’re properly focusing your time and resources from the very beginning of the process, to maximise your chances of success.
Why product success is now even more difficult to achieve
Right now, the world is caring for an ageing population and a new set of health challenges. The demand for cheaper and more usable diagnostics and treatment options is growing fast. Meanwhile, advances in miniaturised tech and AI are offering unprecedented opportunities for developers and disruptors alike.
Demand is strong and developers are responding with new and exciting solutions. Different competitors are bringing wearables, apps and other innovations to market. But there’s risk at every turn.
Just consider the following.
- Medical device production cycles have become 50% faster over the last five years
- Software failures are now the number 1 reason for medical device product recalls
- Medical device recalls increased by 71% between 2016 and 2019
To succeed in this market, therefore, medical device developers must:
- Understand from the outset precisely what kind of device they are developing and how they will meet stringent regulation and quality standards
- Plan an integrated approach to design, development and manufacture to manage risk in a frictionless way
- Focus on implementing a Quality Management System (QMS) that can help them deliver differentiated, user-centric design as flexibly and rapidly as possible.
This section of our website is designed to help you prepare for these requirements. To help you realise the scale of the task before you, plus the kind of tools you’ll need to navigate the design, development and regulatory challenges that lie ahead.
.webp?width=750&name=med-dev-intro-img2%20(1).webp)

.webp?width=750&name=med-dev-sec-1-img1%20(1).webp)
.webp?width=750&name=med-dev-sec1-info1%20(1).webp)


.webp?width=750&name=med-dev-intro-img1%20(1).webp)
.webp?width=750&name=med-dev-sec2-info2%20(1).webp)
.webp?width=750&height=590&name=Medical%20Device%20Development%20Design%20Control%20(1).webp)
.webp?width=750&name=med-dev-se2-img4%20(1).webp)
.webp?width=750&name=med-dev-2%20(1).webp)





%20(1).webp?width=133&height=76&name=ISO%20IEC%2027001%20(1)%20(1).webp)