
What if there were a digital Quality Management System that could be exactly right-sized to your needs?
Cognidox is an electronic document management system that provides a flexible, digital framework for ISO 13485 and FDA 21 CFR 11 compliance. It offers all the controls, tools, and features you need to store, index, and structure your documentation to meet regulatory requirements.
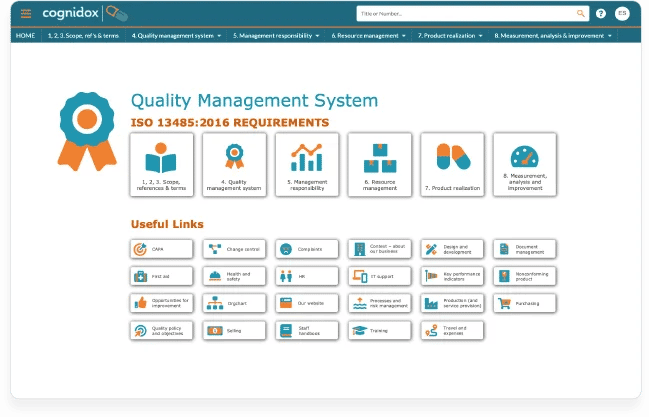
With Cognidox, you can build your ISO-compliant QMS as a fully featured intranet site, providing an accessible and intuitive navigation for your team.
Under the intranet layer is a powerful electronic document management solution (eDMS) into which you can upload your existing quality documents or add them as they are generated.
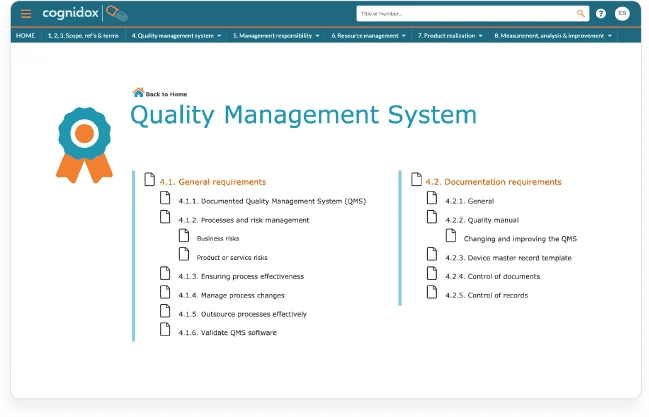
Cognidox brings all the powerful document controls you would expect and that are required by the FDA and ISO 13485:
As and when your workers or auditors need to understand your procedures for dealing with CAPAs, complaints, change control, and every other regulated activity, they can be just a click away from a flowchart (built out in Visio) that shows exactly what is required of them.
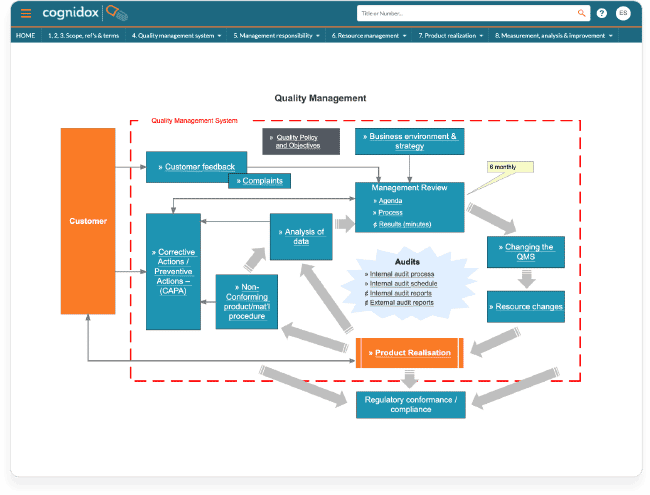
With a graphical eQMS, your whole organisation and external auditors can see you have created the required, rigorous design process, with regular checks against user needs and provision for verification and validation at key stages.
The graphic has deep links to the locations of the specified controlled files related to user requirements and designs. By the end of your development process, you’ll have compiled the full ‘Design History File’ required by the FDA.
.webp?quality=low&width=650&height=495&name=Quality%20management%20system%20product%20development%20(1).webp)
No need to struggle to create usable and accessible forms to capture feedback and issues from internal and external sources.
The Cognidox eQMS provides the custom forms and reports to cover these (and other scenarios):
These forms are part of the solution but are flexible enough for you to edit as you need to or use as the basis for adding other forms. You can create drop-down menus, labels, free-form boxes and the like.
How do you show auditors that training has taken place, that your team is competent, and that changes in process or regulation have been understood?
With Cognidox, GxP training management is fully integrated into your QMS. No separate tools. No disconnected records. Just an automated, compliant training system that works as part of your document control processes.
Cognidox makes it easy to:
You can define job-specific learning paths, automate certification, and build up proof of training over time - with no extra tools or duplicated admin. Whether you’re meeting ISO 13485, FDA QMSR, or EU MDR, Cognidox delivers the evidence you need to prove your team is trained, competent, and inspection-ready.
With Cognidox, eQMS validation is made simple. We help you meet the requirements of ISO 13485, FDA 21 CFR Part 11, and EU MDR, using a structured approach that supports your internal processes.
Whether you are building your validation file from scratch or updating for a new release, Cognidox gives you the documentation, traceability, and test evidence you need.
Cognidox provides the tools to support both traditional IQ, OQ, PQ and CSA (Computer Software Assurance) validation methods:
You stay in control of the process. We provide the structure and support.
For those businesses that want to keep control over their own processes but still save the time of designing and documenting a QMS completely ‘from scratch’, Cognidox’s digital framework is the answer.
It’s a system that can cut out the wasted time of a manual review and approval process, stripping out unnecessary bureaucracy from the way you work, and keeping you laser-focused on producing the documentation you really need to drive your product forward.
Receive instant blog updates, straight to your inbox
Copyright 2026 Cognidox Ltd | Company Number: 06506232 | VAT number: GB 930970714