 In 2008, the ICH Q10 Pharmaceutical Quality System (PQS) guidelines introduced a comprehensive model for an effective QMS in the pharmaceutical industry that could supplement GMP (good manufacturing practice) requirements. But why did the industry so desperately need a new paradigm in their approach to quality management and what will it require for your company to adopt them?
In 2008, the ICH Q10 Pharmaceutical Quality System (PQS) guidelines introduced a comprehensive model for an effective QMS in the pharmaceutical industry that could supplement GMP (good manufacturing practice) requirements. But why did the industry so desperately need a new paradigm in their approach to quality management and what will it require for your company to adopt them?
What Is ICH Q10 and Why Does It Matter?
The ICH Q10 guidelines are a recommended model for building a Quality Management System for the pharma industry. They are the brainchild of the ICH (International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use).
The ICH Q10 model is based on the ISO 9001 quality standard and is designed to be used together with regional Good Manufacturing Practice (GMP) requirements to create a holistic system of continuous quality assurance and improvement specific to pharma developers.
Why the Pharma Industry Needed ICH Q10
The cost of quality in pharmaceutical development has always been high. However research shows problems with governance and quality management are still dogging the speed of product development and profitability for many drugs companies.
McKinsey’s recent report indicates that drug development costs and timelines continue to rise, with the estimated average cost of bringing a drug to market now at $2.6 billion. That’s a 140% increase over the past ten years.
Their report suggests that a holistic transformation in drug development process, including improved quality and compliance, is necessary to bring medicines to market faster, reduce development costs, and enhance planning and safety decision-making.
Pharma companies have been slow to embrace quality management innovation
Pharmaceutical companies have traditionally been slow to embrace the latest quality management approaches and compliance innovations.
This has led to advice from the FDA for developers to move beyond basic adherence to cGMP (current good manufacturing practice) and look to advanced QMS models to embed “continual process and system improvement efforts” in everything they do.
These kinds of pressures are exactly why the ICH developed their PQS guidelines, so pharma companies could begin to develop and manufacture their products to a robust, risk-based, common standard.
What Does the ICH Q10 Pharmaceutical Quality System Include?
Here’s the ICH Q10 diagram illustrating the scope and focus of the PQS guidelines:
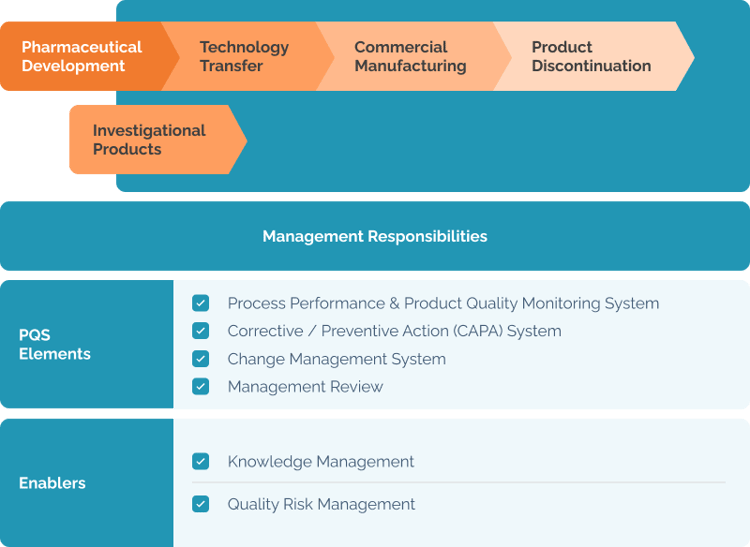
Source: ICH
The diagram shows how the Pharmaceutical Quality System covers all product lifecycle stages. It shows how the PQS complements GMP requirements specific to each country and also applies to investigational products. These lifecycle stages are:
1. Development
Processes and procedures governing how products are invented and developed, including exploratory and clinical development studies.
2. Technology transfer
The transfer of product and process knowledge between development and manufacturing and within or between your operational sites.
3. Commercial manufacturing
Controlling and continually improving the product realisation and distribution stages.
4. Discontinuation
Managing the end of the pharmaceutical product lifecycle in a controlled, consistent way.
The system guidelines define four core ‘pillars’ or quality system elements to support these lifecycle areas:
- Process performance and product quality monitoring
- Corrective action (CAPA) management
- Change management
- Management review
The 4 Pillars of ICH Q10 You Must Understand
Let’s briefly explore the 4 pillars of the pharmaceutical quality system as specified in various clauses of the guidelines:
1. Process Performance and Product Quality Monitoring System:
A definition and focus on the systems and methods used to monitor and control the processes and end products. It involves collecting and analysing data, identifying trends, and using statistical methods to ensure and improve the quality of the pharmaceutical product. This system is crucial for making informed decisions about product quality and process improvements.
2. Corrective Action and Preventive Action (CAPA) System:
This clause of the PQS deals with the systems required for identifying, documenting, and implementing corrective and preventive actions. The CAPA system is a critical component of the PQS, as it helps identify the root causes of detected or potential problems and take appropriate steps to prevent their recurrence.
3. Change Management System:
This clause outlines the approach for managing changes in a controlled and systematic way. It covers changes to processes, equipment, raw materials, and other elements that could affect the product. The goal is to ensure that any changes are evaluated, reviewed, and approved before implementation. This system helps maintain product quality throughout the product lifecycle.
4. Management Review
The clause involves regular management reviews of process performance and product quality. This includes assessing the effectiveness of the PQS, the state of control of processes, product quality trends, and the need for improvements. Management reviews are essential for ensuring continuous improvement and making decisions about product and process changes.
Underpinning the whole PQS are the requirements for a top-down managerial focus on implementing quality systems, knowledge sharing and risk management.
Is ICH Q10 Mandatory?
The ICH Q10 is not a regulatory requirement in itself but rather a model or framework for a pharmaceutical quality system. It's designed to enhance the quality of pharmaceutical products and is aligned with other key quality guidelines, such as ICH Q8 "Pharmaceutical Development" and ICH Q9 "Quality Risk Management."
However, while ICH Q10 is not a mandatory standard, its principles are widely recognised and adopted in the pharmaceutical industry. Regulatory authorities in ICH member regions (the United States, European Union, and Japan, among others) often expect pharmaceutical companies to adhere to these principles as part of their commitment to maintaining high-quality standards in drug development, manufacturing, and distribution.
How to Implement ICH Q10 with a Digital QMS
You can’t implement a PQS to the ICH Q10 standards with a paper-based approach. Likewise, trying to digitise and automate your QMS with a mixture of Google Docs and Excel sheets can be a recipe for disaster.
Digitisation and automation through a proprietary eQMS (electronic Quality Management System) can significantly enhance the efficiency and effectiveness of your process. But companies embarking on a migration to a digital should be mindful of all the potential benefits and pitfalls of shifting to a digitised approach.
Here are some key points to consider:
Streamlining Process
An eQMS can automate and streamline many of the required quality management processes specified in the standard, reducing manual workload and the potential for human error. This includes automating workflows for document control, CAPA processes, change management, and training management.
Data Integration and Accessibility
With an eQMS, all quality-related data can be stored and integrated into a single platform, making it easier to access, analyse, and report on quality metrics for future audits. This centralised data repository is crucial for informed decision-making and effective management review.
Compliance and Traceability
Automation helps in maintaining compliance with regulatory requirements. An eQMS provides traceability and audit trails, which are essential for inspections and audits. It ensures that all actions, changes, and decisions are properly documented and retrievable.
Risk Management
An eQMS can support risk management processes by providing tools for risk assessment and analysis. It can help in identifying, evaluating, and mitigating risks systematically.
Real-time Monitoring and Alerts
With real-time monitoring capabilities, an eQMS can provide immediate alerts for issues or deviations, allowing for prompt corrective actions. This feature enhances the process performance and product quality monitoring system.
Continuous Improvement
An eQMS can facilitate continuous improvement by providing data analytics and trend analysis tools. These tools can help identify areas for improvement and track the effectiveness of implemented changes.
Training Management
An eQMS often includes features for managing these records, ensuring that all personnel have undertaken required training; providing self-certification that documents have been read and courses completed.
Enhanced Collaboration
The right eQMS should improve collaboration across different departments and sites by providing a common platform for sharing information and best practices.
Scalability and Flexibility
As the organisation grows and processes evolve, the right eQMS should scale accordingly, providing flexibility to adapt to changing regulatory environments and business needs.
Cost Efficiency
Over time, the automation and efficiency provided by an eQMS can lead to significant cost savings by reducing the need for manual processes and enabling more effective use of resources.
Why You Need a LEAN and Scalable eQMS
But not every eQMS is created equal. For some scaling companies, shifting to a digital QMS approach can start imposing new and unnecessary bureaucracy on already stretched development teams. ‘Heavy duty’ pharma QMS can come with SOPs built for big, multinational companies, which amount to over-processing for smaller, scaling teams.
For scaling companies, it’s vital to choose a flexible digital QMS that can help you impose required structure and governance without slowing the pace of your work. You should look for SOPs and CAPA forms that are easy to adapt to match how you already work as you look to formalise and automate your quality processes. You don’t want to be forced to change an established process just to satisfy the needs of a software provider.
Final Thoughts: Aligning with ICH Q10 for Long-Term Success
In the words of the FDA’s Dr Woodcock, the aim of driving shared quality standards across the industry is ultimately to build:
‘A maximally efficient, agile, flexible pharmaceutical manufacturing sector that reliably produces high-quality drug products without extensive regulatory oversight’
In the battle to produce a robust and self-sustaining culture of quality in the pharma industry, scaling companies need to find an eQMS solution that is as LEAN and agile as they need to be.
Last updated 20 May 2025





%20(1).webp?width=133&height=76&name=ISO%20IEC%2027001%20(1)%20(1).webp)