
Chip designs are becoming more complex and expensive to develop, verify and validate. Opportunities for fabless developers are multiplying with new government investment and demand from big tech. But complexity and urgency can lead to flawed execution and expensive mistakes. Here’s why every fabless semiconductor company needs to think about governance from the start.
McKinsey says semiconductor companies are the unsung heroes of digital transformation, but the sector has been hitting the headlines in recent years. News of Chip shortages in the Covid crisis and their impact on the car industry, led the general public to understand their central importance to every day life, often for the first time.
The world of the future runs on semiconductors
Right now, semiconductors are propelling the innovations in AI and quantum computing that are poised to revolutionise the world of automotive, medicine, and sustainable technologies once again.
Now the Chips are down
Western governments, for the first time, have become focused on controlling the component supply chains that will power innovation and commercial success in an unstable world.
The Chips for America and the European Chips Act are promising investment in native semiconductor R&D and fabrication infrastructure to protect future supply chains from Chinese control.
At the same time, many tech brands have ambitions to bring chip design and manufacture in-house. Among these is Bosch who have invested billions of Euros in their German foundry. Meanwhile, RISC-V seems to promise a future free from the licensing straight-jackets that big-tech wants to exploit.
Opportunities abound for chip developers
There are opportunities for fabless companies in Europe and America to access funding and bring innovation to the EV, healthcare, defence and countless other industries.
But returns on these investments are urgently required. And no matter how smart these companies and innovators are, they will need to expedite the highest quality designs if they are not to disappoint their investors. With hundreds of millions of dollars potentially at stake, a new level of governance will need to be achieved.
Fabless semiconductor companies can burn bright and crash
In the past, many semiconductor firms have burnt bright but been unable to deliver, as innovation got bogged down in ever more complicated iterations. Whole years could be lost in development blind allies. Sprawling documentation and silioed teams made approval processes inconsistent and prone to error. Talent shortage also slowed down development - requiring more dispersed teams to meet demand without the collaborative software to support it. Botched tapeins and tapeouts could introduce multi-million dollar errors into the production process.
How to avoid project failure
Semiconductor start-ups are increasingly advised (and often required) to adopt document control software or eQMS (electronic quality management system) platforms at the outset of their development to streamline process, collaboration and meet the necessary regulation (such as ISO 9001, ISO 26262 and ISO 13485).
7 reasons you need an eQMS to control your chip design project
1. Chip design is super expensive - the cost of verification and validation is high
International Business Strategies (IBS) recently released projections about 2nm-class chip design. They estimate the development cost of a relatively large 2nm chip to be $725 million.
“Software development and verification accounts for the lion's share of chip design development costs — approximately $314 million for software and about $154 million for verification.”
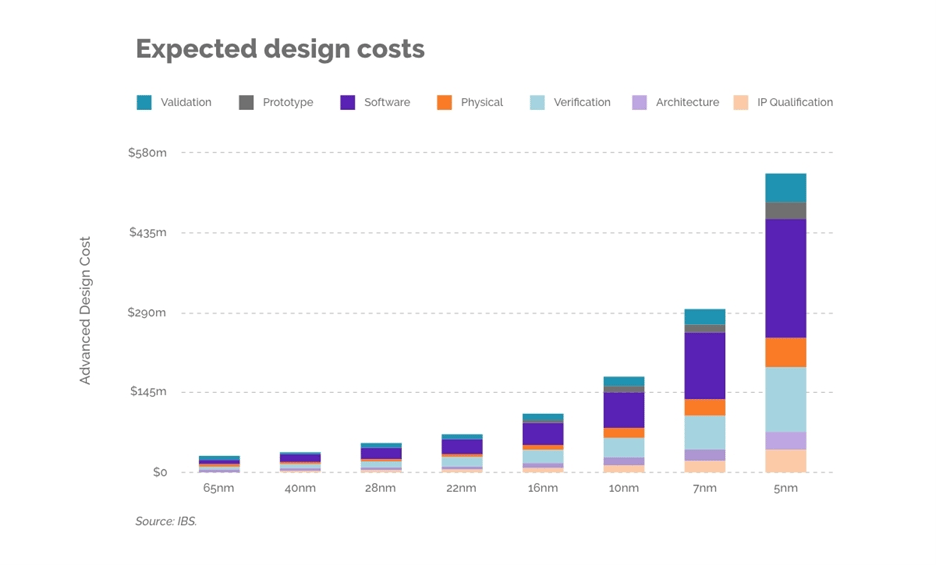
That’s a lot of money riding on process.
Formal verification and validation process where deliverables are approved against designs in a continuous cycle of ongoing quality checks, require software more powerful than a One-Drive/Excel mash-up to get right.
Choosing the right eQMS platform will you give you the tools you need to collate and compare documentation in the right sequence, ensuring sign off by the right people at the right moment before the next phase of your process can be triggered.
2. Version control is paramount
As designs evolve, multiple versions of schematics, layouts, and other critical documents are created. Without a robust document control system, it's easy to lose track of the most recent or correct version, leading to potential design flaws and costly reworks.
3. Your IP is at risk
Chips often incorporate proprietary technologies and intellectual property (IP). A properly managed eQMS system built to ISO 27001 standards will give you the level of internal access control and protection against hacks your team needs to guard against critical data loss.
4. Regulatory compliance
The semiconductor industry is subject to various regulations, especially when designing chips for specific sectors like automotive or healthcare. An eQMS system with the right document control tools ensures that all designs meet the most stringent quality standards like ISO 26262 (for driverless tech) and ISO 13485 (for medical devices), with the paper trail to prove it.
5. Collaboration
With many stakeholders involved in chip design, from architects to engineers and testers, effective collaboration is essential. And in the midst of a global talent shortage, we need the digital tools to support a truly cross-border approach.
The right document control systems can provide a central platform where team members share, review, and update documents in real time wherever they are in the world.
6. You need audit trails
In case of discrepancies or failures, it's vital to trace back the design and decision-making process. eQMS software provides a clear audit trail, detailing who made what changes and when, bringing quicker resolution and accountability.
7. You need to expedite delivery while managing new risks
By streamlining access, ensuring version accuracy, and facilitating collaboration, document control within a QMS can significantly reduce the time it takes to bring a chip from concept to market.
And right now AI is also set to speed up much of this iterative design process for semiconductors. But this output still must be documented, stored, indexed and made available for audit. As AI tools help to collapse timelines, there will be novel governance challenges like this to negotiate - particularly to prove compliance for components destined for driverless cars or medical devices.
Companies will need to choose eQMS platforms that can help impose rigour, planning and appropriate risk assessment - particularly important for components destined for healthcare and automotive clients where the highest standards of traceability apply.
Conclusion
Semiconductor development is an expensive, time-consuming process - and many start-ups fail to stay the course and get their product to market. But in our experience, those companies who do are those who take governance and quality management seriously from the beginning.
Choosing the right eQMS platform that can scale with your needs is key to developing the new generation of semiconductors that the market is demanding. They can give you a digital framework for meeting the most stringent compliance requirements, while helping you accelerate your route to market.
In this highly competitive space, flexible document control is at the core of the quality management process you will need to adopt.




%20(1).webp?width=133&height=76&name=ISO%20IEC%2027001%20(1)%20(1).webp)