Writing clear, compliant Standard Operating Procedures (SOPs) is key to quality management in regulated industries. Whether you’re preparing for ISO 13485 audits or looking to streamline internal training, these 8 expert tips will help you document SOPs that are actually used — not just filed away.
There are many reasons why organisations need to document their SOPs. From ensuring uniformity in end products to training staff and meeting regulatory demands. Good SOPs are the key to quality assurance and sharing organisational knowledge efficiently. But how do you actually write them so they have maximum impact?
What is SOP documentation?
A Standard Operating Procedure (SOP) document records the way key tasks in your organisation should be carried out. A formal SOP is helpful for any job that needs to be repeated in a particular way to maximise efficiency and eliminate product non-conformities. Quality standards such as ISO 13485 and ISO 9001 require that critical SOPs are clearly documented and made easily accessible to the people who need to use them as part of their everyday work.
Here are 8 tips to follow to ensure your SOP documentation is as effective as possible.
1. Define the Purpose of the SOP
Creating Standard Operating Procedures is never about documentation for ‘its own sake’. So before you start writing, think about why you are generating the document and for whom. Do you need to ensure conformity in some kind of repetitive task? Or is it a more complex process you are documenting? Do you need to ensure some specific GxP or ISO requirements are met in the structure of the task? Are the instructions to be used by your own team or a third-party supplier? What kind of language should you use so that it can be understood by them? Is it likely to be read on a phone, tablet or laptop, If so, how can you ensure it will always be rendered properly on these devices? If the SOP requires detailed instructions to be given, what's the best format to clearly convey that information?
2. Use a consistent format 2. Follow a Standard SOP Format
A typical SOP template might include:
- The purpose and intent of the SOP document - i.e. why it was created
- Its scope - Who and what should be governed by the SOP - (note that the purpose and scope should remain distinct)
- Who does what – List all the different job roles and subtasks required to complete the process defined in the SOP.
- Create step-by-step description of the task – Ask yourself, can it be understood by its target audience or followed by someone who has never done the task before?
- Important references – List and link to related documents as clearly as possible - in a way that supports the intention of the SOPs. This might include providing links to appropriate standards and regulations, approved versions of other and third-party documentation (such as instruction manuals)
- Assumptions – List any assumptions made in the drafting or execution of the written procedure as well as any caveats or warnings that might be necessary for understanding how and when a task should be tackled.
- Materials & Equipment - List materials the user will need to satisfactorily complete the procedure as well as the equipment they will need to do so.
3. Keep SOPs Concise and Task-Focused
SOPs that are over-long can be confusing and impractical for both teaching and learning. Think of it from a training point of view. If a SOP incorporates a long list of subtasks, then this might be an indication that it should be broken down into separate SOPs.
“Overprocessing” is a real risk here. As Shaun Knight, Senior Consultant Quality Specialist at the Cambridge Design Partnership told us:
“A regulation might have four or five bullet points for a given topic which you are required to demonstrate compliance with through your QMS. By having procedures which require well beyond the given regulation, you are not only over-processing and doing more work than is required, but you have given yourself a higher risk of non-compliance due to the size of your documented process.”
4. Collaborate with the People Doing the Work
The most effective way to decide what steps should be included in a SOP is to consult the people who are actually carrying out the work. Get a flip chart and a pen and map it out together using flow diagrams. Let the people who do the task describe to you how the task works. Let them share the moments where things can go wrong or the point at which significant choices have to be made. Together you can create the process maps that can become the SOPs that will be most meaningful and helpful for those who need to use them. If your objective is to optimise the way your business performs the tasks you are documenting - working together in this way is crucial to gain insight and collectively agree on the ‘way you do things’ before you commit to a final version.
5. Use Clear, Practical Language
When documenting SOPs be careful to keep descriptions of tasks concise and unambiguous. You need to strike a balance between giving required details and becoming so prescriptive that following them becomes burdensome. Be precise, not pedantic. Aim to keep the attention and enthusiasm of your audience as they use your SOP to understand what the required outcomes are.
6. Test SOPs with First-Time Users
When you’ve completed your process documentation perform a dry run of the activity using your SOP as your guide. If you can, use an individual from a different team who has never performed this task before and see whether they can reproduce the steps based on your documentation. This will show up any gaps in your SOPs and will allow you to correct any mistakes or make updates to optimise their utility. Validate the process and its outcomes against what was required and intended by the SOP.
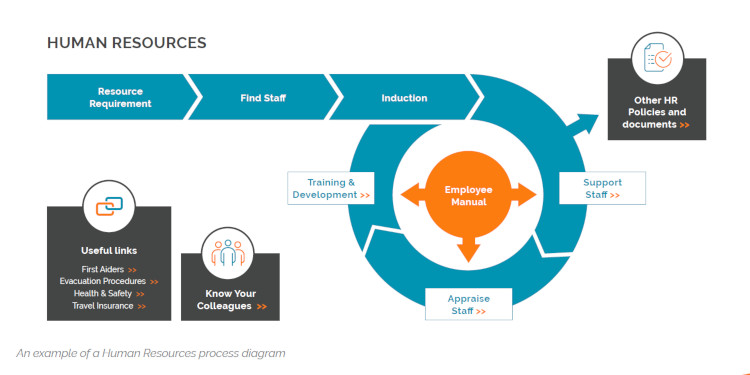
7. Visualise the Process with Flowcharts and Media
The use of flow charts, diagrams, pictures and even videos, in your SOPs can make the difference between SOPs that merely tick compliance boxes and those that are actually helpful and practical for their intended audience. Graphical representations of the flow of tasks are a great way to remove ambiguities, demonstrate dependencies and flag up points where errors can happen or choices need to be made.
%20(2)%20(1).webp?width=750&name=cognidox-gbms-pp-img-2%20(1)%20(2)%20(1).webp)
We would argue that the ability to capture processes graphically and for them to be accessible to the business as a whole should be a key feature of any digital business or quality management system you choose. A Graphical Business Management System, for example, can help you visualise the entire structure of a business diagrammatically, letting you drill down into the structure of individual departments and their SOPs as required. This helps workers contextualise operations while giving them the detail they require to do their jobs more effectively.
8. Review and Update SOPs Regularly
Your work isn’t finished once the standard operating procedure is written. In order for it to remain relevant and useful, it’s got to be updated to reflect changes in the way you do things, the advent of new equipment and new regulatory requirements. Storing and maintaining SOPs in a version-controlled document management system will help you keep a historical record of the evolution of your business practice, which will be helpful both for troubleshooting and auditing purposes. The right Quality Management tools will even be able to notify key stakeholders at regular intervals to review the SOPs for accuracy and relevance.
The last word
SOPs should be more than just box-ticking for compliance, they should be an essential resource. To help this happen we would advise the following. Always keep the needs of your intended audience in mind when you’re documenting your SOPs. And see that the documentation remains accessible, up-to-date and relevant.
A formal SOP should support your GxP or ISO 13485 requirements while remaining usable day-to-day. Use this SOP compliance checklist to make sure your documentation hits the mark.
If you can manage that, there’s a greater chance your documents will prove useful, and be used across your organisation.
Blog post updated on 14 April 2025





%20(1).webp?width=133&height=76&name=ISO%20IEC%2027001%20(1)%20(1).webp)